Prof. Jo Chang-shin developed non-fluorinated electrolytes!
페이지 정보

작성자 최고관리자
댓글 0건 조회 684회 작성일 2022-11-11 17:30
본문

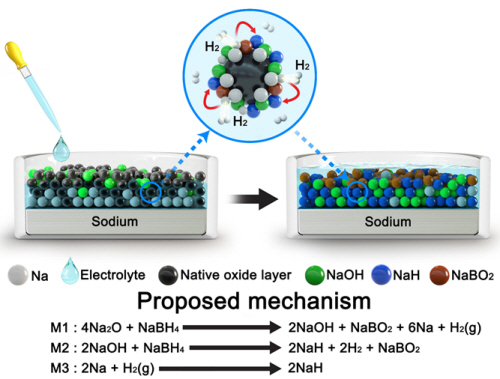

Professor Jo Chang-shin of POSTECH GIFT and Professor Lee Jin-woo of KAIST Biochemical Engineering conducted joint research to develop a non-fluorinated electrolyte for long-life sodium (Na) metal cathodes and high-power seawater cells.
Fluorine (F) has greatly contributed to improving the cycling endurance and power density (“electrochemical performance”) of batteries and has become an essential element of various next-generation battery electrolytes in addition to the currently commercialized lithium-ion batteries. However, a non-fluorinated electrolyte to replace it is essential because F is expensive and toxic to the human body and the environment. The joint research team designed a F-free electrolyte that could replace the existing fluorinated electrolyte to achieve electrochemical performance that exceeds those of the fluorinated electrolyte, which remaining price-competitive. Inspired by a recent report that sodium hydride (NaH) can replace NaF, the research team designed an electrolyte dissolved in an organic solvent (including ether, C-O-C bonds) using boron hydride (NaBH4) salt. Boron hydride is a reducing agent and is widely used in industries that require organic and inorganic synthesis. In addition, the research team applied this electrolyte that uses NaBH4 (NaBH4-electrolyte) to seawater cells; and at a high current density of 1 mA cm-2, the NaBH4-electrolyte achieved survived more than 150 charge/discharge cycles the existing fluorinated electrolyte survived about 35 cycles. Furthermore, NaBH4-electrolyte achieved a power density of 2.82 mW cm-2, whereas the fluorinated achieved only 2.27 mW cm-2, so the research team's NaBH4-electrolyte is expected to contribute to the commercialization of seawater cells by reducing their cost and extending their life. The study was published in the 15th issue of October's 10th volume of the international journal Energy & Environmental Science, and was selected as the outside back cover. (Thesis: Designing Fluorine-Free Electrolytes for Stable Sodium Metal Anodes and High-Power Seawater Batteries via SEI reconstruction)
- 이전글Prof. Sang-Min Lee was selected as the regional innovation mega project for the Secondary Battery field. 23.04.21
- 다음글GIFT Soohyun Kim, Dong Won Lee won the Award at the 2022 Fall Conference of the Korea 22.11.24
댓글목록
등록된 댓글이 없습니다.



